Bet Vs Langmuir Surface Area When to Use Which Multilayer
We start by looking at the adsorption process and mono-layer form. 2there is no interaction between each adsorption layer.

Results Obtained From Bet Analysis A Nitrogen Adsorption Isotherms Download Scientific Diagram
𝑎𝑎 mass of sample.

. The base area nothing but the surface area of adsorbent required to arrange them in monolayer Langmuir is larger than the base area. Langmuir adsorption model considers that only a monolayer of adsorbate can be formed on top the surface. In 1916 Irving Langmuir presented his model for the adsorption of species onto simple surfaces.
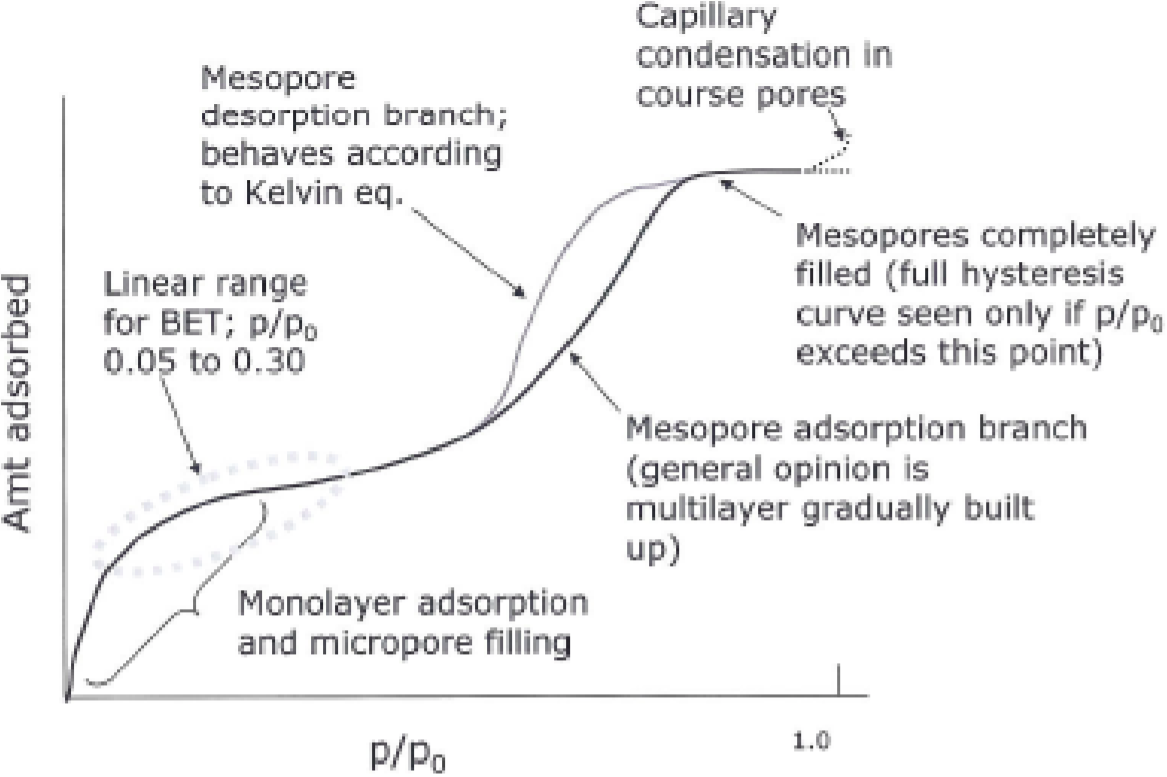
Then if as generally occurs multilayers are formed BET is able to consider that they are not being formed by directly covering the surface but by covering extra layers. This model represents the multilayer adsorption of the molecules to the adsorbent surface and the assumptions of the BET isotherm are the same as for the Langmuir isotherm model. Ideally five data points with a minimum of three data points in the PP 0 range 0025 to 030 should be used to successfully determine the surface area using the BET equation.
There are a few differences but the main one is that Langmuir can only be used for surfaces which are cover by only one layer of gas and the BET surface area is. Seminar paper from the year 2013 in the subject Geography Earth Science - Miscellaneous grade. 𝑉𝑉 molar volume of adsorbed gas.
BET Surface AreaPorosity AnalyzerGas Pycnometer Helium Abosulte Density EquipmentHigh Pressure Volumetric Analyzer LangmuirBETGibbsBJHMPSFDRDAHKT-Plot. BET method is universally recognized and the most commonly used way for nitrogen adsorption surface area analysis. The specific surface area is usually calculated by using BET theory from the gas adsorption isotherm data.
F-Sorb 2400CE adopts speed and efficient continuous flowing gas nitrogen adsorption principle is an expert for contrast reference materials method single and multi-point BET method and Langmuir method analysis. Brunauer Emmett and Teller BET in 1938 are the first to develop a theory to account for this multilayer adsorption and the range of validity of this theory is approximately between 005 and 035 of the vapor pressure. It is a modification of the Langmuir theory which besides monolayer adsorption also considers multilayer adsorption.
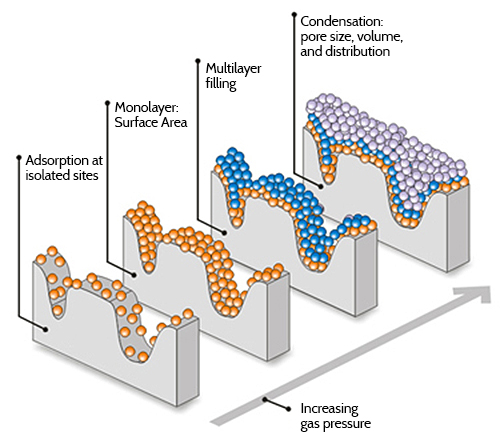
The adsorption sites heavy dots are equivalent and can have unit occupancy. Using a known value for the amount of surface area occupied by each molecule in the monoloayer the surface area of the sample is obtained. BrunauerEmmettTeller BET theory aims to explain the physical adsorption of gas molecules on a solid surface and serves as the basis for an important analysis technique for the measurement of the specific surface area of materials.
This isotherm model is widely used for the adsorption processes in the gassolid systems and the surface area and the porosity of the materials can be determined by. At relative pressures higher than 05 there is the onset of capillary condensation and at relative pressures that are too low only monolayer formation is occurring. The model of BET theory is that the adsorptive starts to adsorb on the strong energy sites on the surface first and then as the pressure is increased the adsorptive adsorbs on the next energy level sites.
The specific surface area it is a physically and chemically important parameter of porous materials. 𝐵𝐵𝐸𝐸𝑅𝑅 𝑆𝑆 𝑡𝑡 𝑎𝑎 m 2 g 𝑆𝑆 𝐵𝐵𝐸𝐸𝑅𝑅 specific surface area. Langmuir was awarded the Nobel Prize in 1932 for his work concerning surface chemistry.
Adsorption is thought to be localized the surface is homogeneous and non-porous lateral interaction is disregarded. 23 RWTH Aachen University Lehr- und Forschungsgebiet Geologie - Endogene Dynamik course. Single point BET analysis For applications where the surface areas of the samples vary but the substance remains unchanged there is a the possibility of using only a single point on the BET isotherm.
In contrast BET model considers multilayer formation. I have done BET analysis to measure surface area and pore sizes. 1gas molecules physically adsorb on a solid in layers infinitely.
I want to convert mmolg into cm3g. In this video we present the calculation of BET surface area from gas adsorption isotherms. For multilayer adsorption the BET model assumes about the same position as that of Langmuir its monolayer counterpart.
The observations are very often referred to as physical adsorption or physisorptionIn 1938 Stephen Brunauer Paul Hugh Emmett and. And 3the Langmuir theory can be applied to each layer. The obtained results showed the adsorption-desorption of N2 in mmolg.
𝑠𝑠 cross-sectional area of adsorbed gas molecule. The most commonly used method for determining the specific surface area is the so-called BET method which obtained its name from three Nobel prize winners Brunauer Emmett and Teller 1938. He hypothesized that a given surface has a certain number of equivalent sites to which a species can stick either by physisorption or chemisorption.

Comparison Of The Langmuir And Bet Isotherms A Langmuir Isotherm B Download Scientific Diagram

49 Questions With Answers In N2 Adsorption Science Topic

Langmuir Theory And Isotherms Type I And Ii Youtube

2 3 Bet Surface Area Analysis Of Nanoparticles Chemistry Libretexts

Multilayer Adsorption An Overview Sciencedirect Topics

Bet Surface Area Ppt Lasopapicture

Direct Method For Evaluation Of Bet Adsorbed Monolayer Capacity Sciencedirect
100 Nano Stories Bet Surface Area By Carlos Manuel Jarquin Sanchez Medium

The Iupac Classification Of Adsorption Isotherms For Gas Solid Download Scientific Diagram

The Bet Adsorption Multi Layer This Model Assumes That For Each Download Scientific Diagram

Comparison Of The Langmuir And Bet Isotherms A Langmuir Isotherm B Download Scientific Diagram

What Is The Difference Between Langmuir And Bet Isotherm Compare The Difference Between Similar Terms

Iupac Classification Of Adsorption Isotherms Typical Bet Range Is Download Scientific Diagram

Adsorption Isotherms Freundlich Adsorption Isotherms Langmuir Adsorption Isotherms Bet Adsorption Isotherms

Multilayer Adsorption An Overview Sciencedirect Topics
A N 2 Adsorption Desorption Isotherm And B The Fitting Curve Of The Download Scientific Diagram

Comments
Post a Comment